Research progress and new thoughts for high-value utilization of low-rank coal according to the future “dual-carbon” policy
-
摘要:
我国低阶煤储量丰富,主要以燃烧利用为主,附加值低;热解、气化等热转化方式一定程度上实现煤的清洁利用,但未改变煤炭能源利用属性;制备碳点等高附加值碳纳米功能材料,可实现煤的高值化和清洁高效利用,但其通常以高阶煤为原料并采用强酸氧化法制备,产生二次污染同时造成残煤资源浪费. 随着国家能源储备转型及“双碳”政策推进,清洁高效利用低阶煤、开发低阶煤非能源属性温和转化利用方法、制备高附加值新型碳纳米材料,成为低阶煤高值化利用的创新举措. 本文简要介绍了我国低阶煤分子结构特性及高值利用现状,分析了煤的利用转化方法和碳纳米材料制备的基本工艺,重点阐述了低阶煤化学转化过程中的氧化方法与最新进展,对比了不同氧化方法效果和不同煤种化学氧化产物、技术及经济性差异,指出未来低阶煤制备碳纳米材料应集中的研究方向为煤结构精准解析、高效解聚转化机理、杂原子赋存形式及作用分析和潜在应用领域拓展.
Abstract:There are abundant reserves of low-rank coals in China, which are primarily used for combustion applications, but they have low-added value. Thermal conversion methods, such as pyrolysis and gasification, have achieved some degree of clean utilization; they do not fundamentally alter the energy utilization properties of coal. The preparation of high-value carbon nanomaterials such as carbon dots, nanodiamonds, and graphene from coal can enable the high-value, clean, and efficient utilization of coal. However, these high-value materials are typically produced using high-rank coal as the raw material and strong-acid-oxidation methods that generate secondary pollution and waste valuable residual coal resources. Following the transformation of national energy reserves and the advancement of the “dual-carbon” policy, development of mild-conversion methods based on the structural characteristics of low-rank coal, aiming to utilize its non-energy properties, can enable the clean and high-value utilization of low-rank coal. This approach focuses on the preparation of new high-value carbon nanomaterials, and represents a major innovative strategy for the high-value utilization of low-rank coal. This review focuses on the latest progress and basic principles of mild-oxidation conversion, utilization of low-rank coal, and the preparation of carbon nanomaterials, including vapor deposition, arc discharge, microwave, laser, and various chemical oxidation methods. Regarding the chemical oxidation method, the basic principles and progress of oxidation with different oxidizing media (such as air, nitric acid, H2O2, and oxidative salts) are introduced. Among the different oxidation methods, H2O2 has been evaluated for its environmental friendliness and high efficiency, and can be studied further as a key technology for the preparation of low-rank coal materials. This study compared and analyzed the preparation techniques of materials using different coal types via chemical oxidation methods, demonstrating the product, technical feasibility, and economic viability of mild-oxidation methods to prepare carbon nanomaterials from low-rank coal. The authors recommend that future research on the preparation of carbon nanomaterials from low-rank coal should focus on four aspects: (1) accurate analysis of coal structure, and understanding the evolution of coal structure and its derived carbon at the molecular level; (2) efficient depolymerization and transformation mechanism; by utilizing the activity of low-rank coal and the structure of oxygen-containing organic functional groups, we can achieve efficient conversion of low-rank coal and improve yield and economy by developing reasonable processes, increasing in-situ characterization or calculation combinations; (3) analysis of heteroatom occurrence forms and functions, study the genesis characteristics of complex minerals and heteroatoms in low-rank coal and their influences on the structures and properties of coal-based carbon materials, and develop functional carbon-based composite materials with unique structures; (4) expansion of potential application areas, conduct research on the preparation and application of coal-based carbon materials, focus on the development of cost-effective and scalable synthesis technologies, and construct multifunctional, high-performance, coal-based functional carbon materials. It is hoped that the proposed work and its findings will provide a reference for the coal conversion industry and the high-value utilization of carbon nanomaterials.
-
低阶煤指褐煤、长焰煤、不黏煤、弱黏煤等变质程度较低的煤,具有高水分、高活性(含氧官能团多)、高挥发分、低热值、低含碳量、易燃易碎等特性. 我国低阶煤储量丰富,约占我国煤炭资源总量的55%,其利用方式主要有燃烧、热解、气化和液化等,其中燃烧占比最大[1]. 热解、液化、气化可以改变煤中碳质成分形态,如制备成焦油、半焦、柴油和液化气等高值产品. 但本质上能源属性未发生改变,且过程中存在高能耗、工艺复杂以及建设成本昂贵等问题[2−3]. 随着国家能源储备转型及“双碳”政策推进,开发低阶煤的非能源属性利用方法和制备高附加值的新型碳纳米材料,成为低阶煤利用的创新举措.
以碳点为代表的纳米功能材料由于其可调的尺寸和表面/边缘结构、丰富的官能团、优良的化学反应性,在环境、催化和储能等领域具有应用潜力[4]. 煤作为最丰富的天然碳质材料之一,其结构中含有大量的微晶石墨结构,易发生剥离和化学改变,因此被认为是理想前驱体[5]. 本论文结合低阶煤清洁高效利用的国家战略需求和我国低阶煤资源禀赋特征,针对低阶煤结构、氧化解聚现状及碳纳米材料制备方法进行综述,旨在通过对不同氧化方法和制备工艺对比分析,对煤的高效转化及碳纳米材料制备进行总结,为低阶煤的高附加值利用奠定理论基础.
1. 低阶煤结构特性
煤结构是煤开发利用的基础,了解煤的结构,对煤的高效、精准利用及煤炭产业链延伸具有重要意义. 尽管不同变质程度的煤在成分和性质上均有所差异,但有机质组成元素均为C、H、O、N、S等元素,且内部含有丰富的芳香族和脂肪族等有机质. 具体而言,其结构模型普遍包含基本结构单元(如苯环、萘环等),这些基本结构单元通过桥键(如—CH2—、—O—等)、官能团(如—COOH、—OH等)和主要烃基构成的侧向取代基连接而成[6]. 迄今已有130多种煤的大分子结构模型,但总的来说,随着煤变质程度的升高,煤中芳香结构增加而脂肪结构不断减少,基本结构单元的芳香环数也有类似的规律. 对于变质程度最高的无烟煤,其化学结构更趋近于石墨结构[7].
煤结构模型始于简单抽提实验分析[8],其仅包含92个原子,由脂肪链和交联的芳香环组成,主要用于分析简单煤炭转化,或用于理解煤结构差异. 随着先进表征技术的进步,物理技术方法对煤结构进行直接测试分析,具有准确真实、客观便利等优势. 如X射线衍射可获取微晶结构信息;固态核磁共振谱可获得各种碳结构比例及含量等,这些物理表征技术为煤的全面分析提供了多角度的信息,已被煤化学领域的专家广泛认可. Jia等[9]研究了三种不同变质程度的煤结构,构建并优化其分子结构模型. 结果表明,随着煤化程度的加深,煤分子中含氧官能团比例逐渐降低,长脂肪链缩短并减少,芳香性增强. 表1为不同类型低阶煤的化学结构模型. 可以看出,随着时间发展,低阶煤的结构模型中不仅考虑了煤中芳香结构单元,还引入了脂肪碳链、杂原子等,并采用分子动力学等不断优化,低阶煤结构的复杂性和完整性得以不断发展.
表 1 不同类型低阶煤的主要化学结构模型Table 1. Main chemical structure model of different types of low-rank coalModel Time Type Characteristics of model Disadvantages Fuch and Sandhoff [10] 1942 Bituminous Composed of a large honeycomb-shaped condensed aromatic ring, with oxygen-containing functional groups randomly distributed on the edge No sulfur-containing structure; the types of oxygen-containing functional groups are not comprehensive Wiser[11] 1975 Bituminous Mainly based on aromatic structures, cross-linked by ether bonds, and thioether and short alkane bonds; the sulfur atom is introduced for the first time Stability of surface functional groups; aliphatic structures and condensed aromatic rings in three dimensions are not considered Wender[12] 1976 Lignite Consists of a single aromatic ring connected by aliphatic side chains Small molecular weight and incomplete structure Yang[13] 1987 Lignite Considers the rich oxygen-containing functional groups and stability Nitrogen, sulfur, and hydrocarbons with more than four carbon atoms are not considered Zhu et al[14] 2023 High-ash, low-rank coal The benzopyrene structure and aliphatic carbon chains (such as methylene) are connected to form a three-dimensional network, which is optimized using Material Studio Strong acid treatment during deashing may affect the structure of organic matter in coal Guo et al [15] 2023 Datong coal The main structures of aromatic hydrocarbons are naphthalene and anthracene; hydroxyl, carbonyl, and carboxyl are the main oxygen-containing groups. Characterizations of heteroatoms and molecular dynamics structure optimization are performed No verification of molecular structure Chong et al [16] 2023 Shanxi long-flame coal Qualitative and quantitative methods are used to analyze coal structures, including information on oxygen-containing functional groups, heteroatoms, etc. Ignores the spatial structure of coal 2. 低阶煤高值利用途径
2.1 热解
热解技术作为煤炭清洁利用的重要组成部分,近年来在能源转化、化工原料生产及废弃物资源化等方面得到广泛应用. 热解过程涉及将煤在缺氧或低氧条件下加热至高温,使煤分解为气体、液体和固体产物,从而实现煤的多重资源化利用. 通过采用控制气氛、压力、添加催化剂或开发热解新工艺等手段可以有效改善热解过程煤自由基反应活度,提高热解效率[17]. Ruslan等[18]探讨了使用Fe–TiO2和Fe–HZSM–5作为催化剂和微波吸收剂,开发低阶煤微波热解方式并研究了过程中关键工艺参数(如反应时间、温度和微波功率)对产品性能的影响. Zhang等[19]探讨了不同温度(500~900 ℃)和预处理方法(机械混合和水热共处理)下煤炭衍生污泥(Coal-derived sludge)与低阶煤的共热解处理,结果表明共热解能提高热解焦炭和氢气产率、减少二氧化碳排放,且水热共处理相对清洁,为煤源污泥和低阶煤的资源化利用提供了新视角.
热解技术作为煤清洁利用的一个重要技术途径,在煤炭的高效转化、资源化利用方面具有重要意义. 然而,其在实际应用中依然面临产物控制、能量利用、副产物处理等瓶颈. 未来,随着催化技术、联合工艺、智能化控制等技术的不断发展,热解技术有望在提高效率、降低成本、减少污染方面实现突破,成为煤炭清洁利用的重要组成部分.
2.2 气化
煤气化技术是现代煤化工产业的龙头,是将煤炭转化成清洁的原料气,继而加工成化工产品的关键所在. 在这一过程中,煤与氧气、蒸汽等反应物发生热化学反应,生成合成气(主要成分为CO、H2、CO2等气体). 除压力、温度、催化剂、煤种等因素对氢气产量的促进作用外,开发气化新工艺、气化与碳捕集技术结合均可实现气化效率提升[20]. Lozynskyi等[21]评估了高频电磁场对煤层加热效率和温度分布的影响,论证了其作为新气化方式的可行性,尤其为低阶煤气化潜力提升提供了新的机遇. Zhang等[22]提出了一种创新的低碳氢气生产方法,通过煤分级气化与化学循环技术结合,有效提升了氢气生产效率并实现了近零能耗的二氧化碳捕集,为低碳煤气化氢气生产技术的发展提供了理论依据.
煤气化技术作为煤炭清洁利用的核心技术之一,具有广泛的应用前景和市场潜力. 然而,技术瓶颈仍然存在,如气化效率、催化剂及气化剂优化、煤种差异等问题,需要通过进一步的技术创新和工艺优化加以解决. 今后煤气化技术将发挥更重要作用,将朝着高效、环保、智能化的方向发展,推动煤炭资源的高效利用和清洁转化,为实现低碳经济和可持续发展提供有力支持.
2.3 液化
煤液化是将煤转化为液态燃料(如合成液体燃料或化学品)的技术,已成为重要的煤炭清洁利用方式之一. 近年来,随着全球对能源结构调整和环境保护的关注,煤液化作为替代石油的一种途径逐渐受到重视. 由于煤是由芳香与脂肪结构单元通过复杂方式连接形成的大分子网状聚合物,其结构极为复杂,进而致使煤直接加氢液化过程反应路径同样较为复杂,目前针对煤的液化反应历程的理解尚不深入. Song等[23]分析了煤直接液化技术中催化剂设计方法,重点探讨了煤液化过程的转化机制、催化剂构建及其提升催化效率的途径,为解决催化剂性能、稳定性及液化过程中的关键问题提出思考. 尽管当前液化技术取得了一定突破,但是转化率低、出油率不足、大量焦化、苛刻的反应条件和高能耗仍影响煤液化反应的进行,催化剂的稳定性和活性是煤液化技术中的关键因素[23]. 尤其是在直接液化中,催化剂容易受到煤中杂质(如硫、氮、重金属等)的影响,导致催化剂中毒和失效. 因此,如何开发高效且耐用的催化剂是煤液化技术中的重要研究方向.
综上所述,低阶煤热解、液化和气化等热处理转化技术,可以实现煤的形态转变,从最初的易自燃、结构复杂、不稳定的“固体形式”转变为“焦炭”、“液体”、“气体”等较稳定能源形式,后续通过加工,实现煤的高效清洁利用,但其能源利用属性未发生根本改变.
2.4 材料化
我国煤炭产业正处于转型和升级的关键时期,随着“双碳”目标的推动,煤炭作为能源燃料属性的比重将逐渐减少. 目前,低阶煤的非能源化利用主要根据其含氧官能团丰富的特性,生产高附加值含氧化合物,如腐殖酸、苯多酸、短链脂肪酸和酚类化合物等,或生产表面活性剂、生物肥料、吸附剂、功能材料等.
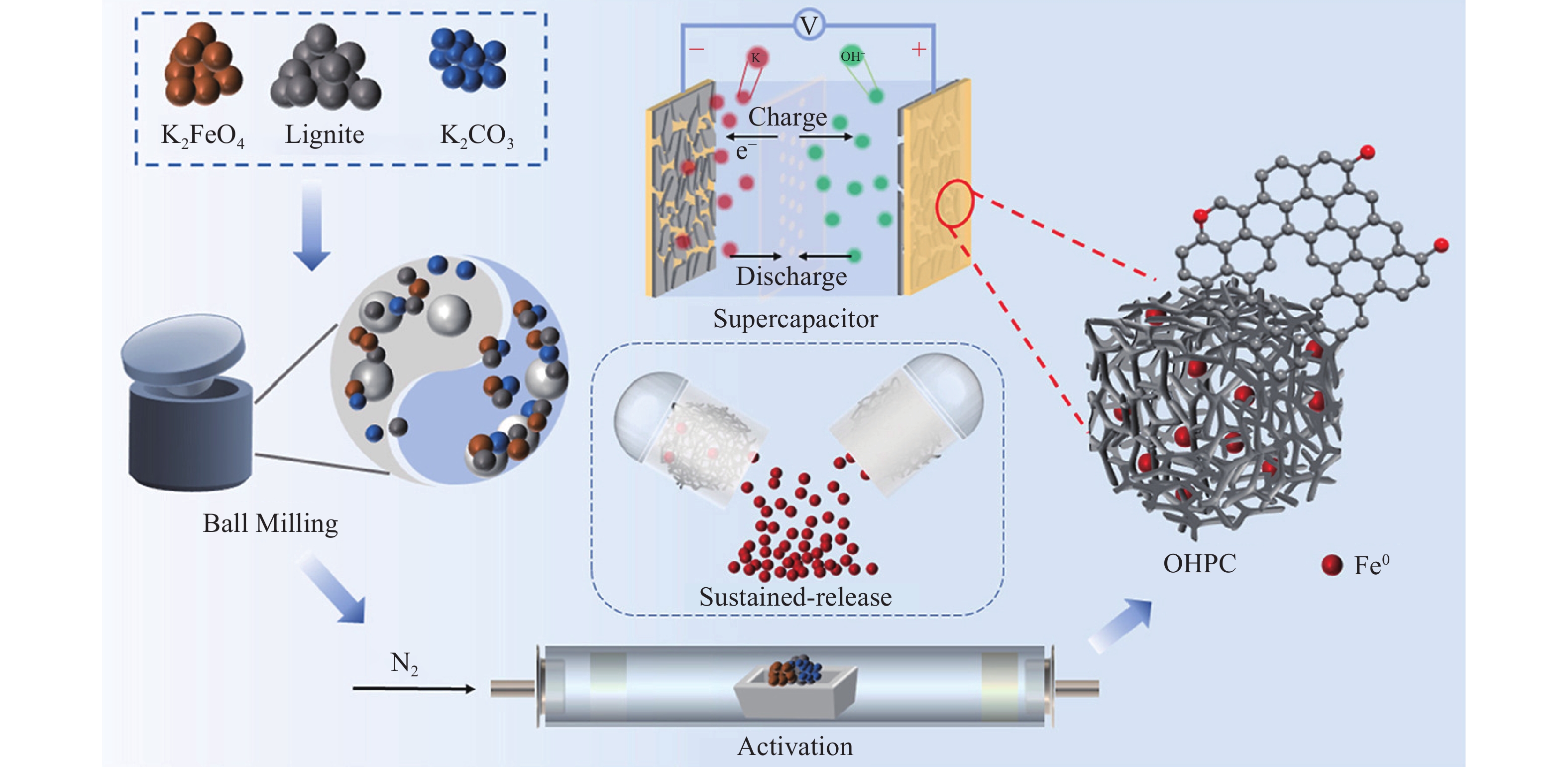
宫贵贞[24]对4种不同变质程度的煤进行研究,通过“分级萃取—柱层析分离—重结晶”技术路线,对煤的氧化转化产物进行精细分离,得到了8种苯多酸甲酯,为从天然大分子中合成苯多酸提供了新的途径. 王知彩等[25]利用褐煤制备腐殖酸并改性,结果表明,改性腐殖酸能显著改善水泥浆的流动性和减水性能. Liu等[26]以褐煤为原料,使用球磨催化热解的方法制备多孔碳材料(OHPC),其制备工艺如图1所示,优化后的OHPC具有较高的比表面积(
1638 m2·g–1)和良好的孔结构,为开发低成本、高性能超级电容器提供了新的途径.除制备以上材料外,利用低阶煤制备纳米材料(碳点、纳米金刚石、氧化石墨烯等)具有高值化、高效、低成本等优势,其主要归因于低阶煤具有纳米尺寸的石墨结构、优异的芳香族结构、脂肪族结构可调性及挥发分高易造孔等特点. Ye等[27]对三种不同类型煤进行处理并通过错流过滤分离,成功制备荧光碳点,通过控制合成温度实现碳点尺寸可控,进而实现碳点荧光发射波长可调,制备的碳点可进一步用于生物成像、传感探针及光电子等领域.
低阶煤材料化利用前景广阔,制备含氧小分子化合物,涉及柱层析技术,后续萃取分离效率较低;制备腐殖酸过程中需用大量酸碱材料,易造成环境二次污染,且经济价值不高;制备活性炭往往需要使用含钾化合物进行活化,此过程需要大量碱,导致成本上升和炭材料收率降低等问题;利用低阶煤制备碳纳米功能材料,能够有效提升低阶煤的经济价值和应用范围,可为煤炭行业的可持续发展提供了新的途径.
3. 煤基碳纳米材料制备
20世纪90年代以来,碳点、纳米金刚石、富勒烯、碳纳米管及石墨烯等相继发现,带给人们一个全新的碳化学视角,并引起碳纳米材料在材料科学、凝聚态物理、化学等学科中的研究热潮. 以煤为前驱体制备碳纳米材料方法包括气相沉积法、化学氧化法、溶剂热法等.
3.1 气相沉积/电弧放电法
气相沉积法(CVD)和电弧放电法(Arc discharge)都利用高温条件下的气相反应过程,促使煤中碳源蒸发重新排列,形成具有纳米结构的材料,如碳纳米管、碳纳米粒子等. 二者都可通过调节实验参数(如温度、气体流量、电流等)来优化碳材料的性能,如提高导电性、表面活性等功能特性,因此它们在能源、催化、储能等领域的应用具有相似的潜力. 然而,二者在制备工艺和材料特性上存在一些显著差异. 气相沉积法(CVD)通过气相化学反应沉积煤基材料,适用于制备具有高质量、高均匀性的碳纳米材料,且沉积过程更加可控,能够精确调整制备的形态和结构[28]. 相比之下,电弧放电法则通过高温电弧蒸发煤中的碳,并在冷却过程中重新排列碳源结构,形成碳纳米材料[29],其工艺相对简单,设备要求较低,但沉积过程可能不如CVD精确,容易产生较多杂质.
3.2 超声/激光/微波
超声法主要利用超声振动过程中产生的高能量将其中的纳米级结晶碳域进行破碎,生成细小的纳米颗粒. 激光在特定位置精确照射时,此区域激发光热反应、光化学反应或光–热–化学反应. 由于局部热效应,激光辐照不会影响周围未被照射的区域,这使得可以构建多样化的微/纳结构,并实现对特定区域的精确碳化控制. 微波加热的原理在于电介质材料在外部电磁场的影响下,内部介质分子发生极化,形成滞后于电场的极化强度矢量. 这种滞后效应导致与电场同相的电流产生,进而在材料内部引起能量耗散. Zhao等[30]使用激光诱导技术对不同煤化程度的煤进行处理,在真空条件下成功制备石墨烯材料,并对石墨烯性能进行表征,结果表明,亚烟煤、烟煤和低变质无烟煤经激光照射后均可形成层数较少的石墨烯;其中由低挥发性烟煤制备的石墨烯材料结晶度最高,芳香层尺寸最大,平均石墨烯层数最小.
3.3 溶剂热
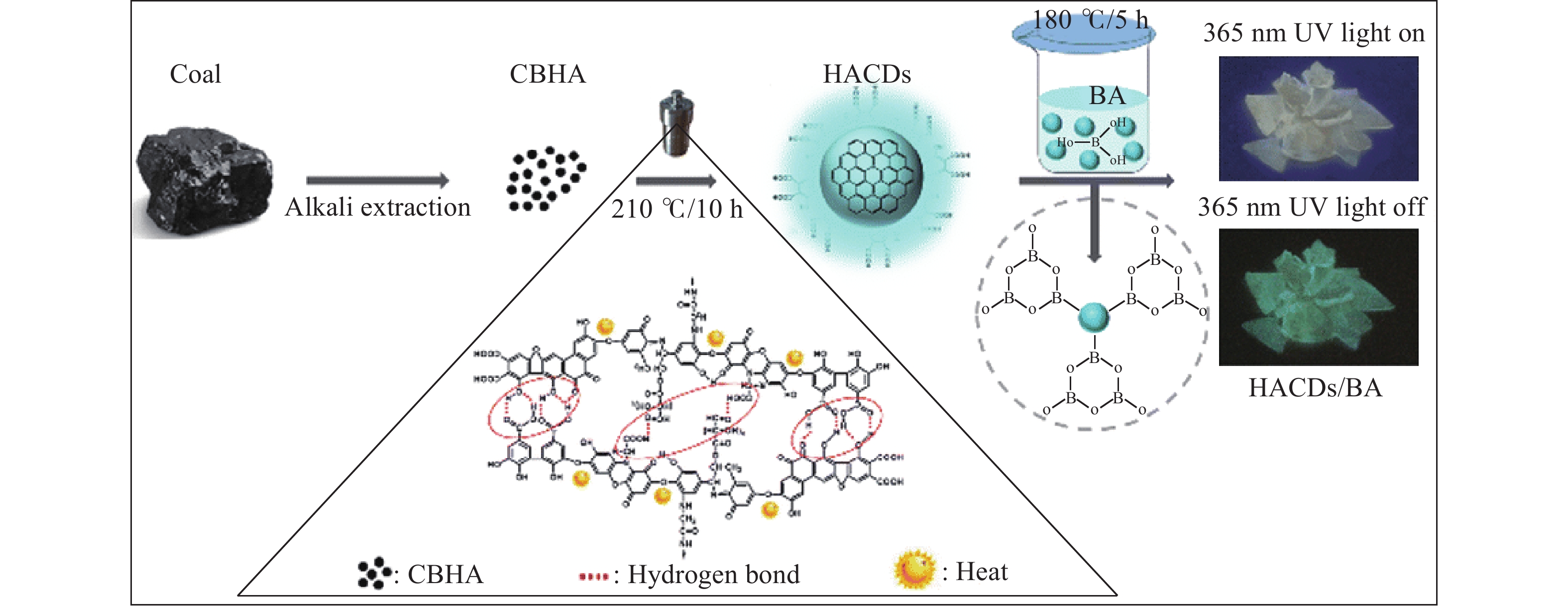
溶剂热法是在水热技术的基础上演化而来,它在高压釜等密闭容器内,使用有机溶剂为介质,在恒定温度和压力下促使原料反应. Xu等[31]使用CO(NH2)2和水在180 ℃条件下在高压反应釜中对煤炭进行处理,并经过改性成功制备光催化分解析氢水的新型复合光催化剂,表现出良好的光催化性能. He等[32]采用溶剂热方法首次制备了在硼酸溶液中可实现室温磷光发射的煤基衍生碳点,其制备工艺与机理如图2所示,在制备过程中,硼酸与碳点在180 ℃条件下发生水热反应生成B—C键,同时由于硼酸热分解产生刚性非晶玻璃态B2O3,导致衍生碳点极易被嵌入刚性衬底中,进一步提高了其室温磷光性能,为制备室温磷光材料开辟了新的方向.
3.4 化学氧化
煤化学中通常认为低阶煤是由晶体碳域经弱键连接的复杂结构,使用化学氧化法可破坏煤晶体碳域间的化学键,保留煤中稳定晶体碳域,此过程还可引入了含氧官能团到制备的纳米碳材料中. 该方法的优势在于它不依赖于特殊的反应设备或条件,从而简化操作流程并降低了能耗.
Ye等[27]使用混酸对三种煤进行处理,发现从烟煤中合成碳点尺寸较无烟煤和焦炭小,碳点尺寸可通过改变氧化温度来调控,这种尺寸调控为石墨烯量子点的应用提供了更多的灵活性和可能性. 本团队使用温和氧化解聚方法对山西长焰煤进行了处理,仅采用H2O2作为氧化剂,通过冷凝回流方法实现煤的高效萃取及碳点高效制备,其制备机理如图3所示,最优参数条件下碳萃取效率为30.86%,制备碳点平均尺寸(1.86±0.62) nm,当碳点水凝胶质量浓度为0.19 g·L–1时其绝对量子产率为2.80%,表现出良好的荧光性能[33],在此基础上,进一步采用有压加碱条件对煤进行深度氧化解聚,碳萃取效率提升至58.54%,实现了碳的高效萃取及碳点高效制备,碳点平均尺寸(1.36±0.42) nm,具有明显双峰荧光发射现象[34].
综上所述,化学氧化法具有操作简单、能耗低、设备简单、产品质量均匀等优点,但当前多以高阶煤为主要原料,且氧化剂通常采用强氧化性酸,易产生废水、造成二次污染的同时残煤无法利用,针对低阶煤含氧官能团多、反应性好的结构特性,“顺势而为”开发温和氧化转化方法制备碳纳米材料具有广阔的应用前景.
3.5 不同煤种制备碳纳米材料方法比较
表2为不同处理工艺及方法制备煤基碳材料的对比,从表中可以看出,不同制备方法和原料,合成碳纳米材料产物及性质均不同. 从表中可以看出,当前不同方法制备碳纳米材料均具有其特定,如气相沉积法制备产品质量高,微波法节能、激光法可精确控制等. 相比其他方法,化学氧化法制备碳材料产品均匀性好,且具有操作简单、高效,设备要求低等优势,并具有大规模生产的潜力. 结合低阶煤的结构及现有温和氧化解聚措施,选择绿色温和氧化剂对煤进行处理,优化氧化工艺,实现煤的高效萃取分离并制备碳基功能材料,将成为煤炭清洁利用和可持续发展的重要方向. 高阶煤如无烟煤,由于更多的石墨化结构,倾向于制备石墨化碳如石墨烯结构,而煤化程度较低的煤如褐煤、长焰煤等由于其表面官能团丰富,局域石墨化结构,倾向于制备小尺寸石墨烯结构或者富含官能团材料.
表 2 不同方法制备煤基纳米碳材料对比Table 2. Specifics of different methods used to prepare nanocarbon materials from coalMethods Raw material Reaction mechanism Product Advantages Disadvantages Reference Chemical vapor deposition Gas molecules produced by coal decomposition Deposition and growth occur on the catalyst’s substrate (Ni or Co) Carbon nanotubes (CNOs), graphene (GO) Extensively used, easy to mass produce, and high quality of prepared products Expensive raw material requirements, high cost, and difficult subsequent separation [37] Arc discharge Coal-based carbon anode The high-temperature requirement during the discharge process causes the anode to evaporate and rearrange the deposits C60, CNOs, GO Preparation of carbon materials with few defects The discharge process is violent and difficult to control [38] Solvothermal Coal React with organic solvents to destroy the cross-linked structure of coal Carbon dots (CDs) Simple operation, high controllability, low-energy consumption Organic solvents cannot be easily recycled and can easily cause secondary pollution [39] Microwave Coal Cracking weak bonds in coal and promoting free radical synthesis reactions Nanodiamond, CDs High efficiency, energy saving, environmental protection, and reduction of by-products High-equipment cost and thermal instability [40] Laser Coal Local high temperature and structural reorganization Nanodiamond, CDs, GO Regional controllable, precise synthesis High-equipment cost and complex process requirements [41] Ultrasound Coal Cavitation effect, local high temperature and high pressure Nanodiamond, CDs Simple operation, efficient dispersion High-equipment cost and limited scale [42] Chemical oxidation method Coal Direct oxidation and exfoliation of graphite flakes in coal Nanodiamond, CDs, GO, CNOs High efficiency, good sample uniformity, simple operation Oxidants are mostly strong acids and prone to cause secondary pollution; oxidizing ability of H2O2 is weaker, and the oxidation time of O2 is longer [27,43] 4. 化学氧化制备煤基碳材料进展
化学氧化的主要原理在于根据氧化程度不同破坏煤中不同化学键,当氧化程度较弱时,可以提取小分子、腐殖酸和制备碳材料;当氧化程度较强时,可以将煤炭转化为CO2. 目前,煤的氧化方法主要有硝酸氧化、氧化性盐氧化、空气氧化、H2O2氧化等.
4.1 硝酸氧化
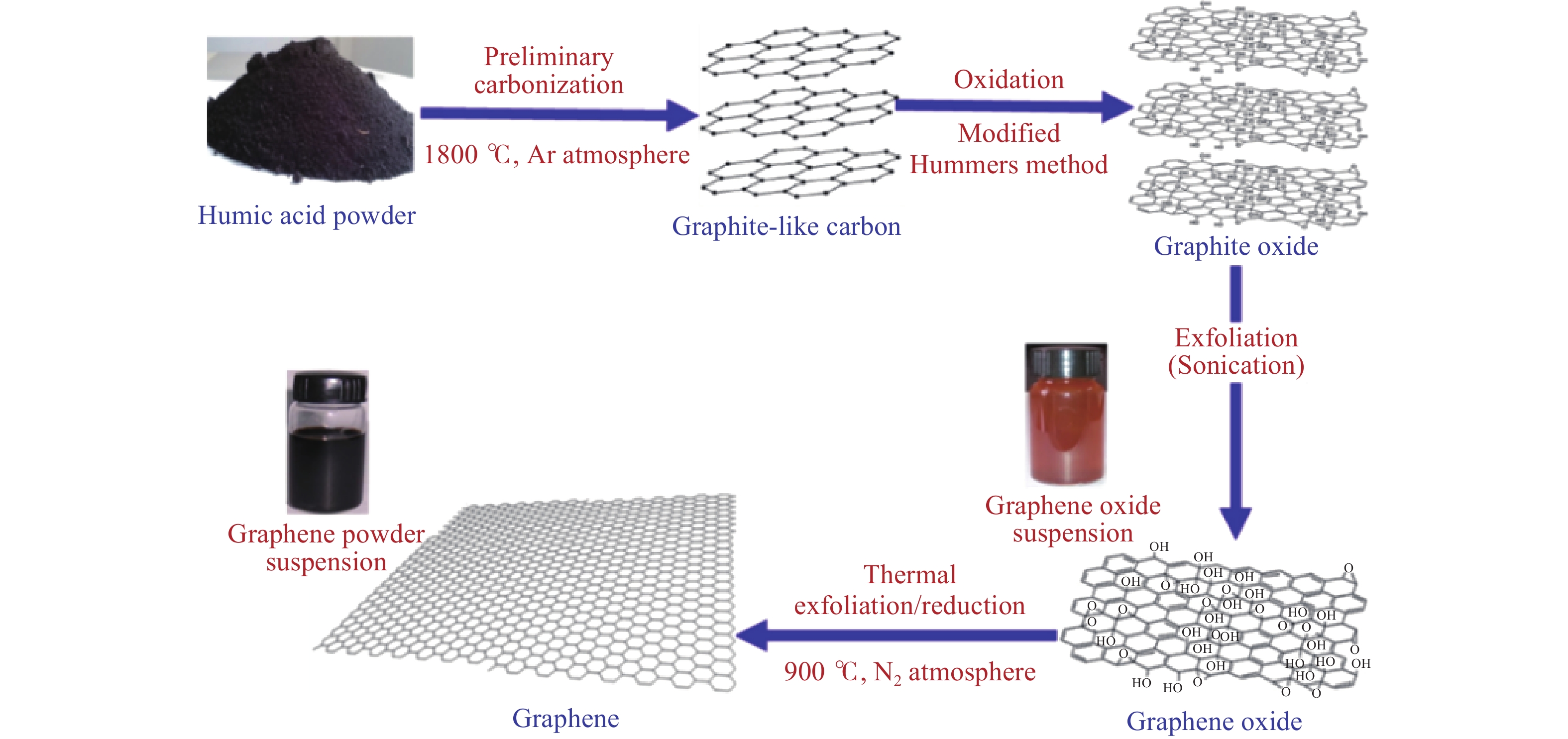
硝酸因氮元素为最高价态(+5)而具有强氧化性. 20世纪60年代,我国科研人员对煤的硝酸氧化制备腐殖酸领域进行了深入研究,并取得了一定的成果[24,44]. 利用硝酸的强氧化性同样可以制备石墨烯材料,如与H2SO4联用的Hummers氧化法,其主要原理为首先将原料转化为硫酸-石墨插层化合物(Graphite intercalation compound),随后氧化剂扩散进入石墨层间(Graphite galleries)产生氧化石墨中间相,最后氧化石墨中间相与水发生反应生成氧化石墨烯[45]. Xing等[46]使用改良后的Hummers方法对煤基腐殖酸进行处理制备腐殖酸,其主要流程如图4所示,首先将煤基腐殖酸进行高温石墨化处理,随后在超声条件下使用两种氧化性强酸对石墨结构进行插层和氧化处理,以减少石墨片层间范德华力并进行分离,最后在还原气氛下进行热处理制备石墨烯. 基于“氧化剥层”的研究思路,刘佳薇[47]使用硝酸及硫酸混合液,在冷凝回流条件下对烟煤进行处理,成功制备直径约1.59 nm碳点,产率为19.96%,具有丰富的官能团和缺陷结构,表现出良好的荧光性能.
总的来说,硝酸氧化具有较好的氧化效果及较高的转化率,可以有效制备碳点、石墨烯、腐殖酸材料等,但其存在环境污染,氧化过程中产生大量废水等问题,且产生含N残渣等. 因此针对性的改进或开发氧化工艺、减少或替代硝酸的浓度或使用量,降低其对环境的污染将是硝酸氧化处理煤的重要发展方向.
4.2 氧化性盐氧化
氧化性盐因强氧化能力和低成本特性,在工业应用中被广泛应用. 其中,次氯酸盐、重铬酸盐和高锰酸盐等最为常见. 次氯酸钠(NaClO)水溶液不仅氧化活性高、经济性强、其对环境的影响较小,具有良好的环境相容性,可以通过电化学方法进行有效回收,将其用于煤的氧化受到广泛关注. 重铬酸钠(Na2Cr2O7)和高锰酸钾(KMnO4)由于其强氧化性、化学性质稳定、易于合成,同样可用于煤的氧化. Chen等[48]研究了NaClO对无烟煤和烟煤结构的影响,经氧化后煤中羟基、芳香烃和含氧官能团的含量上升,而脂肪族组分减少. 这种变化增强了煤的亲水性和芳香性,其显著地改变煤的分子和孔隙结构.
当前,氧化性盐主要用于制备小分子化合物,且仅用于为理解不同煤种的氧化行为和产物特性提供参考. 此过程产生大量副产品,如NaClO水溶液氧化煤会产生大量氯代脂肪酸,Na2Cr2O7和KMnO4氧化易产生大量含重金属铬锰等废水,受限于技术发展,当前仍未能建立高效分离和分析方法,因此氧化性盐氧化尚未大规模推广.
4.3 空气或O2氧化
空气或O2来源丰富且成本低,具有环境友好和工业化潜力,吸引了众多科研工作者进行研究. Yürüm等[49]分别在50、100和150 ℃空气气氛条件下对褐煤进行了氧化,发现空气会选择性的氧化煤中脂肪结构,表现出不同氧化效果. 除干法氧化外,Bergh等[50]在180 ℃条件下直接将氧气或空气通入煤的水悬浮液中,在较短时间内(60 min)约有8%的碳转化为水溶性酸小分子化合物,并制备了腐殖酸等产品. 此外,在碱性溶液中更有利于煤的氧化解聚[44]. Wang等[51]发现烟煤在碱性水溶液中可以更充分地被氧/空气氧化,得到高的苯聚羧酸产率,但是过量的碱会导致有机酸产量的降低.
总的来说,干法条件下对煤进行氧化,反应条件简单,但其氧化性和可选择性较弱,一般情况下需要高温、高压等条件才可提高氧化效果. 添加碱可有效提升煤解聚效果,但过程中碱消耗量大,这些限制制约着干法氧化的实际应用. 湿法氧化相比干法氧化氧化效率明显提升,适当加碱可强化煤的氧化解聚,但仍需在较高温高压下进行,反应条件相对选择性较高.
4.4 H2O2氧化
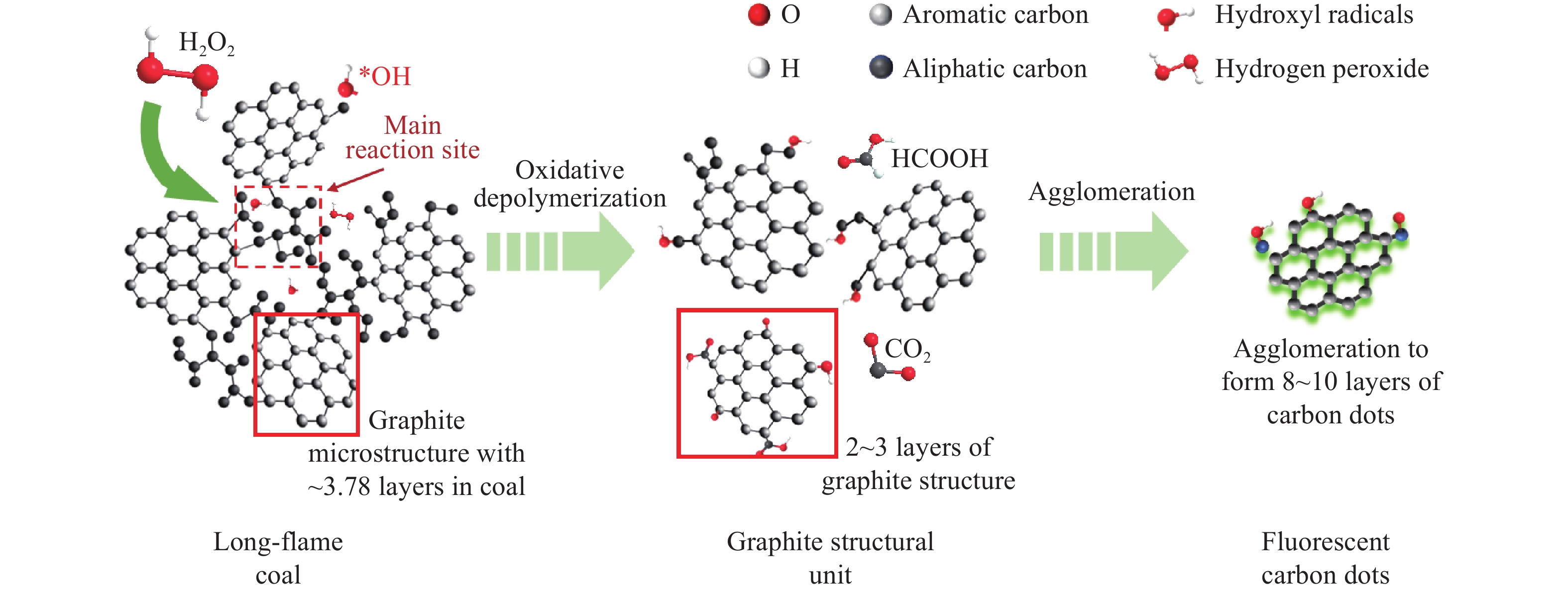
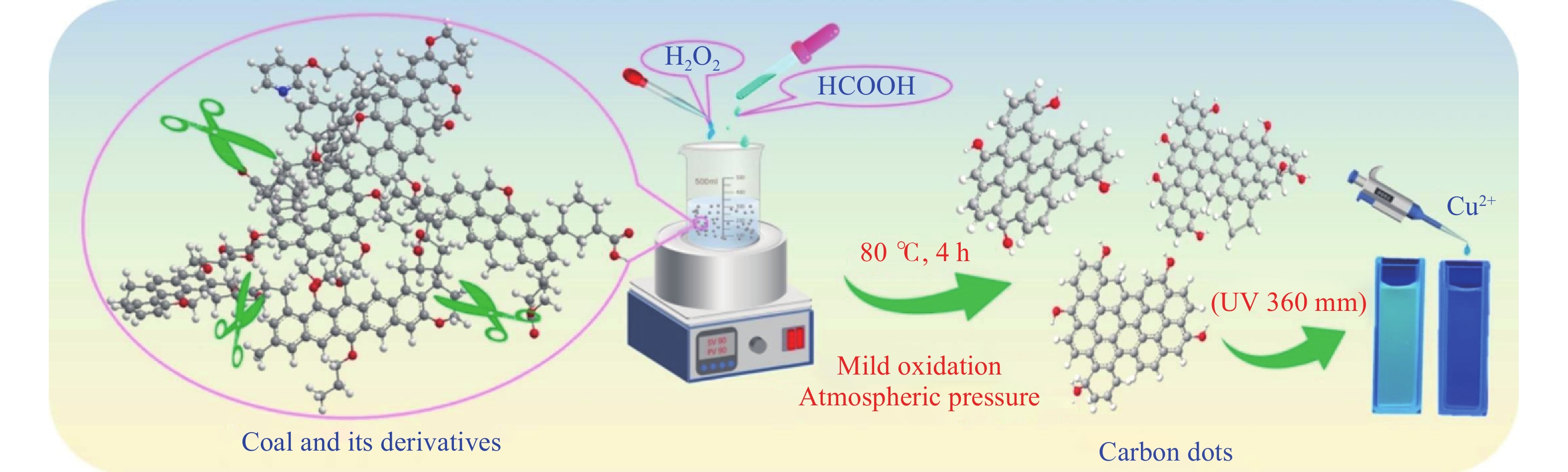
H2O2氧化性强,价格相对低廉、副产物仅为水,具有污染小、操作方便等特点,被广泛用于制备有机过氧化物等材料. 在煤炭领域,使用H2O2用于低阶煤的氧化不仅能实现煤的溶剂抽提率提升,还可以制备小分子含氧酸及纳米碳材料等[24]. 本团队[33−34]使用H2O2对低阶煤进行处理,通过控制液固比、氧化温度等因素控制氧化程度,在冷凝回流条件下成功制备碳点材料及纳米金刚石结构碳纳米颗粒. Bi等[52]使用H2O2和HCOOH混合溶液在常压下处理三种不同煤,成功制备碳点,并用于检测水中的Cu离子含量,其制备机理如图5所示,在制备过程中,甲酸与双氧水协同的强氧化效应,促使煤中石墨晶体碳域间的弱键破坏,继而制备碳点,碳点荧光强度与Cu2+浓度线性相关,据此可进一步拓展其作为探针应用的可能.
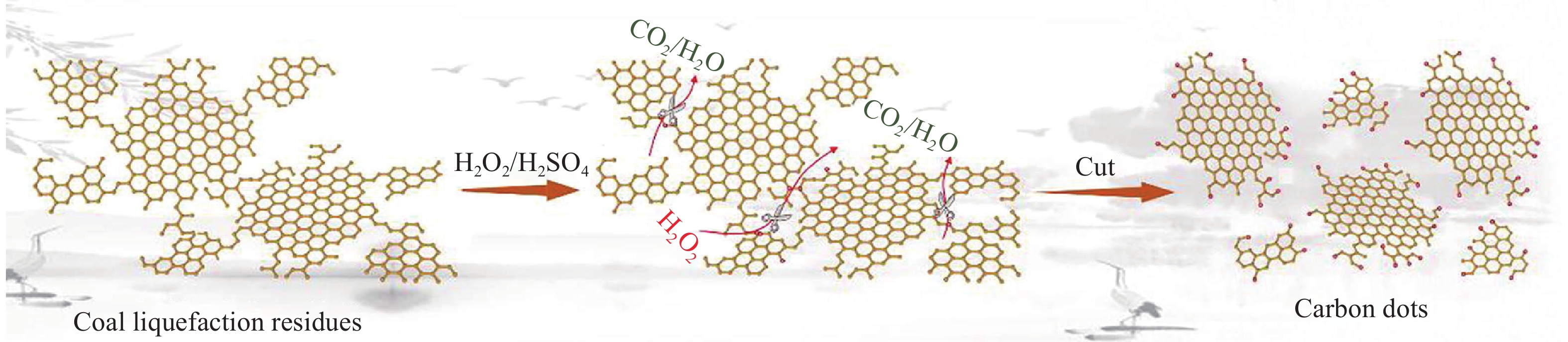
除H2O2与HCOOH溶液混合使用外,使用H2O2与H2SO4混合使用,同样可以达到选择性氧化的目的. Qin等[53]使用温和的氧化方法,以煤液化残渣为碳前驱体,结合过氧酸液(H2O2与/H2SO4)作为氧化剂,制备了功能性碳点,其基本原理如图6所示. 通过催化氢化,改善了液化残渣的氧化反应性,使得煤中的高共轭结构转化为带有脂链的弱连接结构,增强了氧化反应性. 氧化过程中,富含氧基团的碳点在去除灰分的同时形成,进一步通过均匀激活得到比表面积高、密度大的微孔碳,作为超级电容器的高效电极材料. 此外,研究发现碳点作为功能性增强相加入活性炭纳米纤维中,可以显著提高其表面积、机械性能、电导率和超高倍率性能,为煤液化残渣的高效、增值利用提供了新的途径.
综上所述,相比其他氧化方法,H2O2氧化解聚煤具有环保、高效、可控、反应机理简单等优点,一般情况下不要求高温高压、对设备要求较低,其残渣可燃烧、热解利用[35]. 但也有文献报道,只使用双氧水作为氧化剂对煤进行氧化同样可能导致氧化过度,生成CO2逸出从而降低碳的利用率[44]. 添加碱能有效降低多环基团反应能力,从而促进侧链、桥键氧化制备羧酸,且碱性环境能够将氧化过程中生成的羧酸转化为稳定的羧酸盐,防止产物进一步氧化. 基于这一原理,冯波等[54]采用NaOH作为碱性介质,使用双氧水氧化褐煤,高效制备长链脂肪酸和水杨酸. 因此,根据低阶煤结构特性,优化反应条件提高解聚度,制备碳基纳米功能材料,将成为低阶煤高附加值利用的关键研究方向.
4.5 不同煤种化学氧化方法对比
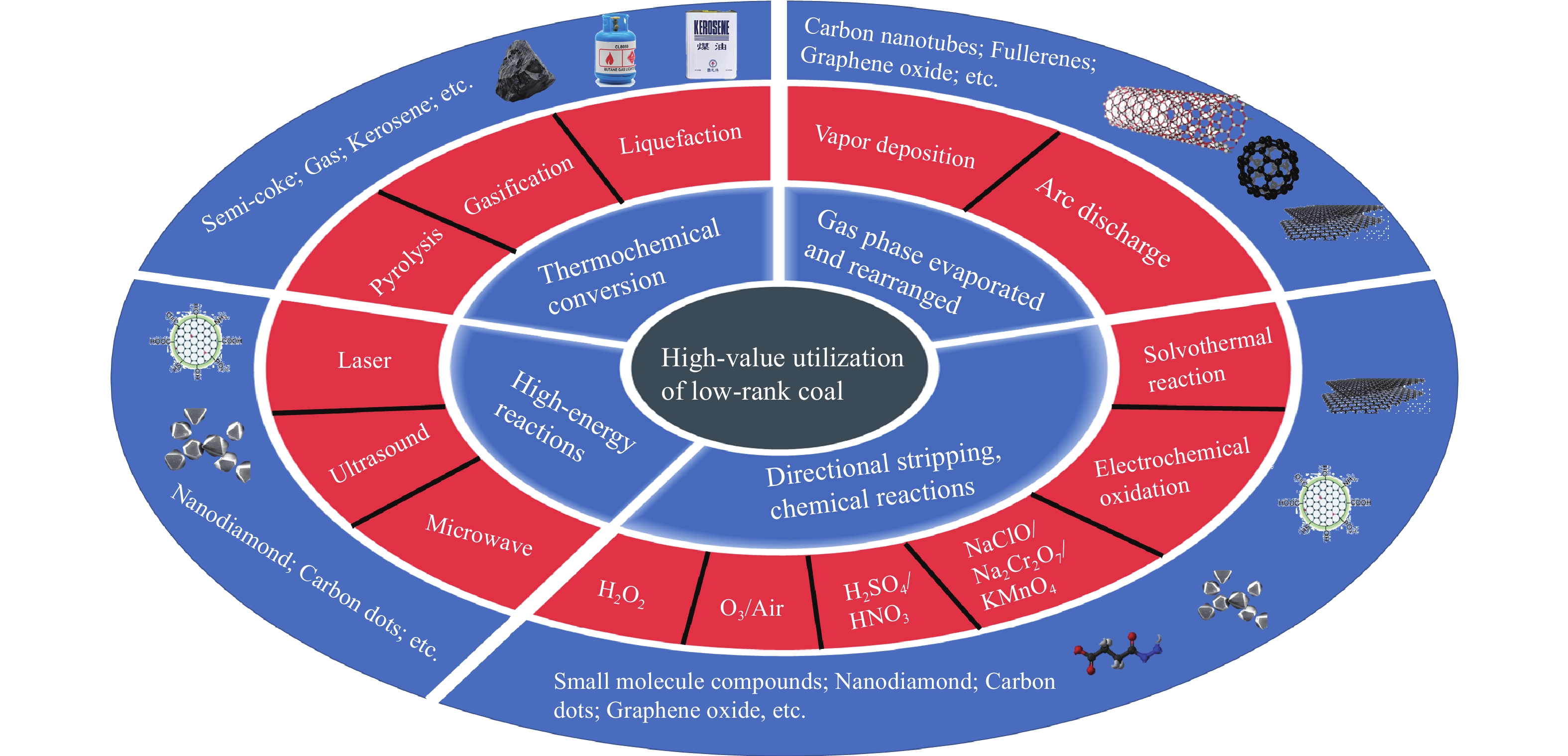
基于不同煤结构分析,低阶煤结构最为复杂,通常具有较高的挥发分和较低的固定碳,表面含有较多官能团,表现出较高的氧化活性. 根据以上材料制备方法进行总结并分析,低阶煤主要高值利用方式及产物如图7所示,可以看出基于热化学转化过程中分子结构裂解并重组,低阶煤可通过热解、气化、液化方式制备半焦、煤气、煤油等产物;利用高温条件下的气相反应过程,通过气相沉积、电弧放电等方法促使煤中碳源蒸发,重新排列可以定向调控产物结构,制备富勒烯、碳纳米管、氧化石墨烯等材料;通过激光、微波、超声等手段利用局部区域高能反应,同样可实现碳纳米材料的定向制备,如制备纳米金刚石、碳点等;相比而言,使用溶剂化学反应,操作较为简单,可实现碳纳米材料的产业化制备,但针对低阶煤较高的氧化活性,避免过度氧化或氧化不足导致的碳质浪费或无法充分材料化的氧化新工艺仍需不断探索. 表3为不同化学氧化方法制备煤基碳纳米材料对比分析,可以看出,化学氧化法制备碳纳米材料主要产物为碳点、小尺寸石墨烯材料等,主要在于化学氧化过程通常无法实现煤结构分子重排. 团队前期研究发现,低阶煤中的石墨微晶结构通过较多亚甲基等弱键连接[16],采用H2O2温和氧化方法可以破坏低阶煤中的弱键,进而剥离微晶结构并制备碳点材料,且该过程可在较低温度下进行. 相比之下,以无烟煤为代表的高阶煤中含有大量石墨结构,单独使用H2O2无法破坏其结构,因此需要开发更强的强氧化方法,如H2SO4或HNO3,以实现无烟煤结构的定向氧化,此过程中要求反应器需具备较强的耐高温和耐酸碱性能,还不可避免的造成二次污染. 但需要注意的是,利用低阶煤制备碳纳米材料过程中较多灰分和矿物质可能对其材料结构及性能有一定影响,在温和氧化过程中,氧化剂对有机/无机嵌布组分处炭/灰界面的调控机制尚不明晰,利用低阶煤制备原位矿相掺杂碳点的应用渠道仍需进一步拓展.
表 3 不同氧化方法制备煤基碳纳米材料对比Table 3. Comparison of different oxidation methods used to prepare carbon nanomaterials from coalOxidation reagents Raw material Product Advantages Disadvantages References H2SO4 Anthracite, bituminous coal, lignite GO Simple method Strong corrosiveness, deashing pretreatment, secondary pollution [55] HNO3 Anthracite, bituminous coal, lignite Multilayer graphene Modifiable, high efficiency High-equipment requirements and secondary pollution [56] HNO3 Coal tar pitch CDs Modifiable, high efficiency High-equipment requirements and secondary pollution [57] Electrochemical oxidation Coal tar pitch CDs The method is simple, mild, and highly controllable Low yield and complex equipment [58] H2O2 Long-flame coal CDs The method is simple, mild, and highly controllable The reaction is unstable and more reagents may be needed [33] O3 Lignite coal CDs Strong oxidizing property, environmentally friendly The reaction is difficult to control and the stability is poor [59] H2O2/H2SO4 Coal liquefaction residues CDs Strong oxidizing property and controllable oxidation degree High-equipment requirements and secondary pollution [60] H2O2/HCOON Coal tar pitch CDs Strong oxidizing property and mild reaction conditions High-reaction rate, corrosive [61] H2SO4/O3 Tar GO Strong oxidizing capacity Risk of excessive oxidation, corrosiveness [62] H2SO4/HNO3 Coal tar pitch Porous carbon Strong oxidizing capacityand low cost Highly corrosive, risk of excessive oxidation [63] 从经济性角度来看,低阶煤作为一种原料成本较低的资源,其制备碳点等碳纳米材料的成本优势明显. 虽然低阶煤制备的碳点纯度相对较低,但通过温和氧化方法,能够有效控制氧化过程,减少对环境的负面影响. 如温和氧化能显著降低废水、废气等二次污染物的生成,从而减少了二次废水处理及其他环境管理的费用. 这一方面可以提高整体经济效益,另一方面也有助于实现更加绿色和可持续的生产过程. 从长远角度来看,随着技术的不断进步,低阶煤的利用潜力将逐渐被挖掘,特别是在高效能、低成本的碳点材料制备及其他碳基材料的生产方面. 通过优化氧化过程、明晰低阶煤中杂原子赋存形式,提升技术水平,低阶煤有望成为一种具有较高性价比的资源,推动相关产业的发展,并实现更好的能源和环境双重效益. 因此,从经济性角度而言,低阶煤的综合利用将有助于降低整体生产成本,提升碳基材料的市场竞争力.
5. 展望
我国低阶煤资源丰富,开发低阶煤制备功能碳材料,不仅对碳减排、低阶煤的高值化利用有着重要意义,更是推进新型煤化工领域和保障国家能源安全的重要举措. 然而,鉴于低阶煤分子结构的复杂性,现阶段低阶煤衍生碳材料的基础研究还相对薄弱,相关研究仅限于合成策略以及物理结构和表面结构的调控. 根据低阶煤现有温和氧化解聚方法,针对晶体碳域丰富的低阶煤进行处理,破坏煤中晶体碳域间弱键并保留煤中微晶石墨结构直接制备碳点,成为低阶煤高值利用和碳点开发新的研究思路.
未来低阶煤制备碳纳米材料的研究应集中在以下方面. (1)煤结构精准解析:需从碳分子工程的角度,研究煤的大分子结构,建立不同类型低阶煤与合成转化过程之间的关系,在分子水平上理解煤结构及其衍生碳的演化机理. (2)高效解聚转化机理剖析:针对复杂低阶煤中碳元素开展定向氧化解聚和产品分离制备碳纳米材料相关研究,充分利用低阶煤活性及含氧有机官能团结构,通过开发合理工艺、增加原位表征或计算结合,实现低阶煤高效转化,提高产率及经济性. (3)杂原子赋存形式及作用分析:研究低阶煤中复杂矿物和杂原子的成因特征及其对煤基碳材料结构和性能的影响,开发具有独特结构的功能碳基复合材料. (4)潜在应用领域拓展:深入开展煤基炭材料的制备与应用研究,重点发展具有成本效益和可扩展性的合成技术,构建多功能、高性能的煤基功能碳材料.
-
表 1 不同类型低阶煤的主要化学结构模型
Table 1 Main chemical structure model of different types of low-rank coal
Model Time Type Characteristics of model Disadvantages Fuch and Sandhoff [10] 1942 Bituminous Composed of a large honeycomb-shaped condensed aromatic ring, with oxygen-containing functional groups randomly distributed on the edge No sulfur-containing structure; the types of oxygen-containing functional groups are not comprehensive Wiser[11] 1975 Bituminous Mainly based on aromatic structures, cross-linked by ether bonds, and thioether and short alkane bonds; the sulfur atom is introduced for the first time Stability of surface functional groups; aliphatic structures and condensed aromatic rings in three dimensions are not considered Wender[12] 1976 Lignite Consists of a single aromatic ring connected by aliphatic side chains Small molecular weight and incomplete structure Yang[13] 1987 Lignite Considers the rich oxygen-containing functional groups and stability Nitrogen, sulfur, and hydrocarbons with more than four carbon atoms are not considered Zhu et al[14] 2023 High-ash, low-rank coal The benzopyrene structure and aliphatic carbon chains (such as methylene) are connected to form a three-dimensional network, which is optimized using Material Studio Strong acid treatment during deashing may affect the structure of organic matter in coal Guo et al [15] 2023 Datong coal The main structures of aromatic hydrocarbons are naphthalene and anthracene; hydroxyl, carbonyl, and carboxyl are the main oxygen-containing groups. Characterizations of heteroatoms and molecular dynamics structure optimization are performed No verification of molecular structure Chong et al [16] 2023 Shanxi long-flame coal Qualitative and quantitative methods are used to analyze coal structures, including information on oxygen-containing functional groups, heteroatoms, etc. Ignores the spatial structure of coal 表 2 不同方法制备煤基纳米碳材料对比
Table 2 Specifics of different methods used to prepare nanocarbon materials from coal
Methods Raw material Reaction mechanism Product Advantages Disadvantages Reference Chemical vapor deposition Gas molecules produced by coal decomposition Deposition and growth occur on the catalyst’s substrate (Ni or Co) Carbon nanotubes (CNOs), graphene (GO) Extensively used, easy to mass produce, and high quality of prepared products Expensive raw material requirements, high cost, and difficult subsequent separation [37] Arc discharge Coal-based carbon anode The high-temperature requirement during the discharge process causes the anode to evaporate and rearrange the deposits C60, CNOs, GO Preparation of carbon materials with few defects The discharge process is violent and difficult to control [38] Solvothermal Coal React with organic solvents to destroy the cross-linked structure of coal Carbon dots (CDs) Simple operation, high controllability, low-energy consumption Organic solvents cannot be easily recycled and can easily cause secondary pollution [39] Microwave Coal Cracking weak bonds in coal and promoting free radical synthesis reactions Nanodiamond, CDs High efficiency, energy saving, environmental protection, and reduction of by-products High-equipment cost and thermal instability [40] Laser Coal Local high temperature and structural reorganization Nanodiamond, CDs, GO Regional controllable, precise synthesis High-equipment cost and complex process requirements [41] Ultrasound Coal Cavitation effect, local high temperature and high pressure Nanodiamond, CDs Simple operation, efficient dispersion High-equipment cost and limited scale [42] Chemical oxidation method Coal Direct oxidation and exfoliation of graphite flakes in coal Nanodiamond, CDs, GO, CNOs High efficiency, good sample uniformity, simple operation Oxidants are mostly strong acids and prone to cause secondary pollution; oxidizing ability of H2O2 is weaker, and the oxidation time of O2 is longer [27,43] 表 3 不同氧化方法制备煤基碳纳米材料对比
Table 3 Comparison of different oxidation methods used to prepare carbon nanomaterials from coal
Oxidation reagents Raw material Product Advantages Disadvantages References H2SO4 Anthracite, bituminous coal, lignite GO Simple method Strong corrosiveness, deashing pretreatment, secondary pollution [55] HNO3 Anthracite, bituminous coal, lignite Multilayer graphene Modifiable, high efficiency High-equipment requirements and secondary pollution [56] HNO3 Coal tar pitch CDs Modifiable, high efficiency High-equipment requirements and secondary pollution [57] Electrochemical oxidation Coal tar pitch CDs The method is simple, mild, and highly controllable Low yield and complex equipment [58] H2O2 Long-flame coal CDs The method is simple, mild, and highly controllable The reaction is unstable and more reagents may be needed [33] O3 Lignite coal CDs Strong oxidizing property, environmentally friendly The reaction is difficult to control and the stability is poor [59] H2O2/H2SO4 Coal liquefaction residues CDs Strong oxidizing property and controllable oxidation degree High-equipment requirements and secondary pollution [60] H2O2/HCOON Coal tar pitch CDs Strong oxidizing property and mild reaction conditions High-reaction rate, corrosive [61] H2SO4/O3 Tar GO Strong oxidizing capacity Risk of excessive oxidation, corrosiveness [62] H2SO4/HNO3 Coal tar pitch Porous carbon Strong oxidizing capacityand low cost Highly corrosive, risk of excessive oxidation [63] -
[1] Ma Z K, Cheng L M, Li L Y, et al. Pollutant emission and attrition performance of low calorific blended coals during co-combustion in fluidized bed. Fuel, 2023, 331: 125782 doi: 10.1016/j.fuel.2022.125782
[2] 刘壮, 田宜水, 胡二峰, 等. 低阶煤热解影响因素及其工艺技术研究进展. 洁净煤技术, 2021, 27(1):50 Liu Z, Tian Y S, Hu E F, et al. Research progress on influencing factors and technology of low-rank coal pyrolysis. Clean Coal Technol, 2021, 27(1): 50
[3] Song H J, Liu G R, Zhang J Z, et al. Pyrolysis characteristics and kinetics of low rank coals by TG-FTIR method. Fuel Process Technol, 2017, 156: 454 doi: 10.1016/j.fuproc.2016.10.008
[4] Hoang V C, Hassan M, Gomes V G. Coal derived carbon nanomaterials—Recent advances in synthesis and applications. Appl Mater Today, 2018, 12: 342 doi: 10.1016/j.apmt.2018.06.007
[5] Du M J, Advincula P A, Ding X D, et al. Coal-based carbon nanomaterials: En route to clean coal conversion toward net zero CO2. Adv Mater, 2023, 35(25): 2300129 doi: 10.1002/adma.202300129
[6] 李瑞青, 唐跃刚, 郇璇, 等. 煤基石墨烯原料与制备技术研究进展. 煤田地质与勘探, 2020, 48(5):1 Li R Q, Tang Y G, Huan X, et al. Progress in the research on the raw materials and the preparation techniques of coal-based graphene. Coal Geol Explor, 2020, 48(5): 1
[7] 张双全. 煤化学. 5版. 徐州:中国矿业大学出版社, 2019 Zhang S Q. Coal Chemistry. 5th ed. Xuzhou: China University of Mining & Technology Press, 2019
[8] Wang J, He Y Q, Li H, et al. The molecular structure of Inner Mongolia lignite utilizing XRD, solid state 13C NMR, HRTEM and XPS techniques. Fuel, 2017, 203: 764 doi: 10.1016/j.fuel.2017.05.042
[9] Jia J Z, Wang D M, Li B. Study on molecular structure characteristic and optimization of molecular model construction of coal with different metamorphic grade. J Mol Struct, 2024, 1295: 136655 doi: 10.1016/j.molstruc.2023.136655
[10] Fuchs W, Sandhoff A G. Theory of coal pyrolysis. Ind Eng Chem, 1942, 34(5): 567 doi: 10.1021/ie50389a010
[11] Wiser W. Hydrogenation of western coal in dilute phase under medium pressures for production of liquids and gases [EB/OL]. Report Online (1975−12−01) [2025−03−04]. https://www.osti.gov/servlets/purl/7192448
[12] Wender I. Catalytic synthesis of chemicals from coal. Catal Rev, 1976, 14(1): 97 doi: 10.1080/03602457608073408
[13] 杨文宽. 腐殖煤的热降解机理和生烃率. 石油与天然气地质, 1987, 8(1):26 Yang W K. Thermo-degradation mechanism and hydrocarbon productivity of humic coal. Oil Gas Geol, 1987, 8(1): 26
[14] Zhu B K, Dong X S, Fan Y P, et al. Structural characterization and molecular model construction of high-ash coal from northern China. Molecules, 2023, 28(14): 5593 doi: 10.3390/molecules28145593
[15] Guo S L, Geng W L, Yuan S J, et al. Understanding the molecular structure of Datong coal by combining experimental and computational study. J Mol Struct, 2023, 1279: 135035 doi: 10.1016/j.molstruc.2023.135035
[16] Chong J K, Cheng X, Xiao L H, et al. Fine characterization of the macromolecular structure of Shanxi low-rank coal. J Mol Struct, 2023, 1273: 134359 doi: 10.1016/j.molstruc.2022.134359
[17] 曹景沛, 姚乃瑜, 庞新博, 等. 煤热解研究进展及其发展历程. 化工进展, 2024, 43(7):3620 Cao J P, Yao N Y, Pang X B, et al. Research progress and development history of coal pyrolysis. Chem Ind Eng Prog, 2024, 43(7): 3620
[18] Ruslan R, Sardi B, Khairuddin K, et al. Microwave pyrolysis of low-rank coal: Enhancing liquid fuel yield and quality using Fe–TiO2 and Fe–HZSM-5 catalysts. J Energy Inst, 2025, 119: 101942 doi: 10.1016/j.joei.2024.101942
[19] Zhang T L, Zhang C X, Ren H, et al. Co-pyrolysis of coal-derived sludge and low-rank coal: Thermal behaviour and char yield prediction. Fuel Process Technol, 2025, 267: 108165 doi: 10.1016/j.fuproc.2024.108165
[20] Matamba T, Iglauer S, Keshavarz A. A progress insight of the formation of hydrogen rich syngas from coal gasification. J Energy Inst, 2022, 105: 81 doi: 10.1016/j.joei.2022.08.001
[21] Lozynskyi V, Falshtynskyi V, Kozhantov A, et al. Increasing the underground coal gasification efficiency using preliminary electromagnetic coal mass heating. IOP Conf Ser: Earth Environ Sci, 2024, 1348(1): 012045 doi: 10.1088/1755-1315/1348/1/012045
[22] Zhang S P, Lv L G, Liu L X, et al. An efficient low-carbon hydrogen production system based on novel staged gasification coupling with chemical looping technology. Energy Convers Manag, 2025, 328: 119625 doi: 10.1016/j.enconman.2025.119625
[23] Song W, Liu P G, Chen X Y, et al. Research progress of catalysts for direct coal liquefaction. J Energy Chem, 2025, 100: 481 doi: 10.1016/j.jechem.2024.09.003
[24] 宫贵贞. 煤在NaOCl水溶液中的选择性氧化和产物的精细分离[学位论文]. 徐州:中国矿业大学, 2011 Gong G Z. Selective Oxidation of Coal in NaOCl Aqueous Solution and Fine Separation of Products [Dissertation]. Xuzhou: China University of Mining and Technology, 2011
[25] 王知彩, 刘伟伟, 周璁, 等. 基于煤基腐殖酸的高效减水剂合成与性能表征. 化工进展, 2023, 42(7):3634 Wang Z C, Liu W W, Zhou C, et al. Synthesis and performance of a superplasticizer based on coal-based humic acid. Chem Ind Eng Prog, 2023, 42(7): 3634
[26] Liu H W, Wang Y Z, Lv L, et al. Oxygen-enriched hierarchical porous carbons derived from lignite for high-performance supercapacitors. Energy, 2023, 269: 126707 doi: 10.1016/j.energy.2023.126707
[27] Ye R Q, Xiang C S, Lin J, et al. Coal as an abundant source of graphene quantum dots. Nat Commun, 2013, 4: 2943 doi: 10.1038/ncomms3943
[28] Wu D, Wang M C, Zeng J W, et al. Preparation and characterization of graphene from refined benzene extracted from low-rank coal: Based on the CVD technology. Molecules, 2021, 26(7): 1900 doi: 10.3390/molecules26071900
[29] 张亚婷, 严心娥, 刘国阳, 等. 煤基石墨烯系列材料的可控制备及其在CO2还原过程中的应用进展. 洁净煤技术, 2022, 28(8):1 Zhang Y T, Yan X E, Liu G Y, et al. Research progress on controllable preparation of coal based graphene series materials and its application in CO2 reduction process. Clean Coal Technol, 2022, 28(8): 1
[30] Zhao Y G, Wang S Q, Wang X L, et al. Coal-based graphene formation by CO2 laser scribing: Various structures and excellent electrochemical performance. Fuel, 2024, 373: 132318 doi: 10.1016/j.fuel.2024.132318
[31] Xu S Y, Li M, Wang Y J, et al. Enhanced photocatalytic hydrogen production from Co–MOF/CN by nitrogen and sulfur Co-doped coal-based carbon quantum dots. J Rare Earths, 2024, 42(5): 838 doi: 10.1016/j.jre.2023.09.020
[32] He Z G, Yuen M, Zhang C, et al. Room-temperature phosphorescence in coal-based humic acid-derived carbon dots. J Mater Chem C, 2024, 12(17): 6333 doi: 10.1039/D4TC00787E
[33] Chong J K, Cheng X, Xiao L H, et al. Green production of tunable multicolor nanoparticles with diamond structure from long-flame coal by a one-step mild oxidation. Ceram Int, 2023, 49(23): 38448 doi: 10.1016/j.ceramint.2023.09.174
[34] Chong J K, Hu G C, Xiao L H, et al. Mild Alkaline-Enhanced depolymerization of Long-Flame coal for the synthesis of Coal-Derived fluorescent carbon dots with application as probes. Fuel, 2024, 369: 131795 doi: 10.1016/j.fuel.2024.131795
[35] 种峻楷. 长焰煤温和氧化解聚制备荧光碳点及残煤利用基础研究[学位论文]. 北京:北京科技大学, 2024 Chong J K. Basic Research on Preparation of Fluorescent Carbon Dots by Mild Oxidative Depolymerization of Long-flame Coal and Utilization of Residues [Dissertation]. Beijing: University of Science and Technology Beijing, 2024
[36] Hu S L, Wei Z J, Chang Q, et al. A facile and green method towards coal-based fluorescent carbon dots with photocatalytic activity. Appl Surf Sci, 2016, 378: 402 doi: 10.1016/j.apsusc.2016.04.038
[37] 侯旺君, 闫翎鹏, 曹哲勇, 等. 煤基零维纳米碳材料的合成、性能及其在能源转换和存储应用中的研究进展. 化工学报, 2022, 73(11):4791 Hou W J, Yan L P, Cao Z Y, et al. Research progress of synthesis and properties of coal-based zero-dimensional nanocarbon materials and their applications in energy conversion and storage. CIESC J, 2022, 73(11): 4791
[38] Awasthi S, Awasthi K, Ghosh A K, et al. Formation of single and multi-walled carbon nanotubes and graphene from Indian bituminous coal. Fuel, 2015, 147: 35 doi: 10.1016/j.fuel.2015.01.043
[39] Shi C, Wei X Y. Microwave-assisted grafting of coal onto nitrogen-doped carbon dots with a high quantum yield and enhanced photoluminescence properties. Molecules, 2024, 29(6): 1349 doi: 10.3390/molecules29061349
[40] Kumar Thiyagarajan S, Raghupathy S, Palanivel D, et al. Fluorescent carbon nano dots from lignite: Unveiling the impeccable evidence for quantum confinement. Phys Chem Chem Phys, 2016, 18(17): 12065 doi: 10.1039/C6CP00867D
[41] Zhang C, Xie Y C, Zhang C, et al. Upgrading coal to multifunctional graphene-based materials by direct laser scribing. Carbon, 2019, 153: 585 doi: 10.1016/j.carbon.2019.07.070
[42] Das T, Saikia B K. Nanodiamonds produced from low-grade Indian coals. ACS Sustainable Chem Eng, 2017, 5(11): 9619 doi: 10.1021/acssuschemeng.7b02500
[43] 李瑞青, 唐跃刚, 罗鹏, 等. 宁夏煤制石墨烯的微观结构演化特征:综合表征分析. 煤炭科学技术, https://kns.cnki.net/kcms/detail/11.2402.td.20240624.1039.001.html Li R Q, Tang Y G, Luo P, et al. Microstructure evolution characteristics of coal-made graphene in Ningxia: Comprehensive characterization analysis. Coal Sci Technol, https://kns.cnki.net/kcms/detail/11.2402.td.20240624.1039.001.html
[44] 陈杰, 王玉高, 王志磊, 等. 低阶煤温和氧化解聚研究现状与展望. 洁净煤技术, 2022, 28(9):133 Chen J, Wang Y G, Wang Z L, et al. Research progress and prospects of mild oxidative depolymerization of low-rank coals. Clean Coal Technol, 2022, 28(9): 133
[45] Garg A, Basu S M, Lee S Y, et al. Simplified one-pot synthesis of graphene oxide from different coals and its potential application in enhancing the mechanical performance of GFRP nanocomposites. ACS Appl Nano Mater, 2023, 6(15): 14594 doi: 10.1021/acsanm.3c03197
[46] Xing B L, Yuan R F, Zhang C X, et al. Facile synthesis of graphene nanosheets from humic acid for supercapacitors. Fuel Process Technol, 2017, 165: 112 doi: 10.1016/j.fuproc.2017.05.021
[47] 刘佳薇. 基于化学氧化法的煤基碳量子点合成及其发光机理研究[学位论文]. 武汉:华中科技大学, 2020 Liu J W. Study on Synthesis and Luminescence Mechanism of Coal-based Carbon Dots Based on Chemical Oxidation Method [Dissertation]. Wuhan: Huazhong University of Science and Technology, 2020
[48] Chen A, Zhai C, Yu X, et al. Quantitative analysis of the effect of NaClO stimulation in anthracite and bituminous coal with FTIR and LF-NMR. Energy & Fuels, 2024
[49] Yürüm Y, Altuntaş N. Air oxidation of beypazari lignite at 50 ℃, 100 ℃ and 150 ℃. Fuel, 1998, 77(15): 1809 doi: 10.1016/S0016-2361(98)00067-2
[50] Bergh J J, Cronjé I J, Dekker J, et al. Non-catalytic oxidation of water-slurried coal with oxygen: Identification of fulvic acids and acute toxicity. Fuel, 1997, 76(2): 149 doi: 10.1016/S0016-2361(96)00194-9
[51] Wang W H, Hou Y C, Niu M G, et al. Production of benzene polycarboxylic acids from bituminous coal by alkali-oxygen oxidation at high temperatures. Fuel Process Technol, 2013, 110: 184 doi: 10.1016/j.fuproc.2012.12.011
[52] Bi Y X, Xing B L, Zeng H H, et al. Eco-friendly sustainable fluorescent coal-based carbon dots as a highly selective probe for Cu2+detection. Fuel, 2024, 378: 132933 doi: 10.1016/j.fuel.2024.132933
[53] Qin F W, Li Q Q, Tang T T, et al. Functional carbon dots from a mild oxidation of coal liquefaction residue. Fuel, 2022, 322: 124216 doi: 10.1016/j.fuel.2022.124216
[54] 冯波, 其鲁, 张敬华, 等. 弱氧化环境下褐煤氧化产物的定性分析. 冶金分析, 2009, 29(1):21 Feng B, Qi L, Zhang J H, et al. Qualitiative analysis of brown coal oxidation products in mild oxidizing atmosphere. Metall Anal, 2009, 29(1): 21
[55] Das B, Kundu R, Chakravarty S. Preparation and characterization of graphene oxide from coal. Mater Chem Phys, 2022, 290: 126597 doi: 10.1016/j.matchemphys.2022.126597
[56] Zhao K, Cao J Z, Qi Y, et al. Synthesis of multilayer graphene and its graphene derivatives from coal. Phys Scr, 2024, 99(3): 035924 doi: 10.1088/1402-4896/ad2327
[57] Singh S B, Dastgheib S A. Characteristics of graphene oxide-like materials prepared from different deashed-devolatilized coal chars and comparison with graphite-based graphene oxide, with or without the ultrasonication treatment. Carbon, 2024, 228: 119331 doi: 10.1016/j.carbon.2024.119331
[58] He M Q, Guo X R, Huang J Z, et al. Mass production of tunable multicolor graphene quantum dots from an energy resource of coke by a one-step electrochemical exfoliation. Carbon, 2018, 140: 508 doi: 10.1016/j.carbon.2018.08.067
[59] Liu X X, Hao J Y, Liu J H, et al. Green synthesis of carbon quantum dots from lignite coal and the application in Fe3+ detection. IOP Conf Ser: Earth Environ Sci, 2018, 113: 012063 doi: 10.1088/1755-1315/113/1/012063
[60] Gong G Q, Liang S J, Shi Y M, et al. Preparation of microporous carbon materials using residual coal from oxidative degradation of lignite as the carbon source and the mechanism and dynamics of its methyl orange adsorption. Colloids Surf A Physicochem Eng Aspects, 2022, 636: 128138 doi: 10.1016/j.colsurfa.2021.128138
[61] Cheng Z F, Wu X Y, Liu L, et al. A highly efficient, rapid, room temperature synthesis method for coal-based water-soluble fluorescent carbon dots and its use in Fe3+ ion detection. New Carbon Mater, 2023, 38(6): 1104 doi: 10.1016/S1872-5805(23)60706-1
[62] Buitrago Sanchez S N, da Silveira Salla J, Cesconeto L P, et al. Synthesis of multi-layer graphene oxide from HCl-treated coke and Brazilian coals by sulfuric acid thermal exfoliation and ozone oxidation. Heliyon, 2024, 10(9): e30546 doi: 10.1016/j.heliyon.2024.e30546
[63] Liu Q, Wu D L, Wang T, et al. Pre-oxidating and pre-carbonizing to regulate the composition and structure of coal tar pitch: The fabrication of porous carbon for supercapacitor applications. Adv Funct Mater, 2024, 34(29): 2400556 doi: 10.1002/adfm.202400556



 下载:
下载: